The scientific interests of Dr. Brankovic’s group are directed towards better understanding of the physical and chemical processes occurring at the electrochemical interface and their use to produce the nanomaterials and nanostructures with novel functionality and application.
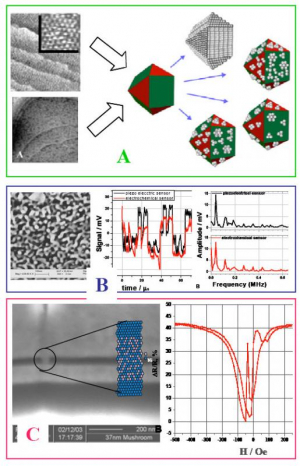
Several examples of the current work are shown in Figure on the right. In the green quadrant (A), the nucleation, growth and self organization of Pt and Pd submonolayers on Au(111) (low-left) and Ru(0001) (up-left) via surface controlled red-ox reaction is studied as the route for design of monolayer electrocatalysts for PEMFC application. In this approach every catalytically active atom counts and, if applied on high surface electrode configuration (nanoparticles), it could lead to design of catalyst materials with high activity and ultimately low noble metal loading.
The blue quadrant (B) shows the nanoporous Au electrode obtained by dealloying process. This structure is studied as model system to demonstrate novel concept where electrical double layer is used as capacitive transducer for ultrasound sensors applications.
In red quadrant (C), the advantages of electrodeposition at nanoscale electrode geometry is used to form magnetic nanocontacts with specific metal-metal oxide structure which could be used as magnetic field sensors for magnetic recording or MRAM applications.
Dr. Brankovic’s research interests include also studies of the additive effect on corrosion and magnetic properties of electrodeposited alloys, electropolishing and its application in ULSIC technology, and the specific aspects of thermodynamics and kinetics of electrochemical thin film growth.
Research Faculty
